Chemistry, Department of: Faculty Series
Patrick Dussault Publications
Accessibility Remediation
If you are unable to use this item in its current form due to accessibility barriers, you may request remediation through our remediation request form.
Document Type
Article
Date of this Version
2009
Abstract
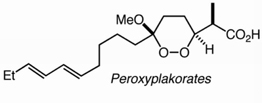
The stereospecific intramolecular alkylation of a hydroperoxyacetal provides the basis for the first asymmetric synthesis of the dioxane propionate core of the peroxyplakorates. Chemoselective hydrometallation of an alkyne in the presence of a peroxide is used to introduce a synthon for the polyunsaturated side chains of the peroxyplakorates. The route suggests a general solution for the 1,2-dioxane unit in many peroxide natural products.


Comments
Published in Tetrahedron 65 (2009), pp. 9680–9685; doi:10.1016/j.tet.2009.09.068 Copyright © 2009 Elsevier Ltd. Used by permission.