Chemistry, Department of: Faculty Series
Robert Powers Publications
Solution structure of Archaeglobus fulgidis peptidyl-tRNA hydrolase (Pth2) provides evidence for an extensive conserved family of Pth2 enzymes in archea, bacteria, and eukaryotes
Document Type
Article
Date of this Version
March 2005
Abstract
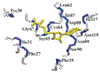
The solution structure of protein AF2095 from the thermophilic archaea Archaeglobus fulgidis, a 123- residue (13.6-kDa) protein, has been determined by NMR methods. The structure of AF2095 is comprised of four α-helices and a mixed β-sheet consisting of four parallel and anti-parallel β-strands, where the α-helices sandwich the β-sheet. Sequence and structural comparison of AF2095 with proteins from Homo sapiens, Methanocaldococcus jannaschii, and Sulfolobus solfataricus reveals that AF2095 is a peptidyltRNA hydrolase (Pth2). This structural comparison also identifies putative catalytic residues and a tRNA interaction region for AF2095. The structure of AF2095 is also similar to the structure of protein TA0108 from archaea Thermoplasma acidophilum, which is deposited in the Protein Data Bank but not functionally annotated. The NMR structure of AF2095 has been further leveraged to obtain good-quality structural models for 55 other proteins. Although earlier studies have proposed that the Pth2 protein family is restricted to archeal and eukaryotic organisms, the similarity of the AF2095 structure to human Pth2, the conservation of key active-site residues, and the good quality of the resulting homology models demonstrate a large family of homologous Pth2 proteins that are conserved in eukaryotic, archaeal, and bacterial organisms, providing novel insights in the evolution of the Pth and Pth2 enzyme families.


Comments
Published in Protein Science (2005), 14:2849–2861. Published by Cold Spring Harbor Laboratory Press. Online at http://www.proteinscience.org/
Since two co-authors are U.S. Dept. of Energy employees, this work is in the public domain.