Chemistry, Department of: Faculty Series
Patrick Dussault Publications
Document Type
Article
Date of this Version
2013
Citation
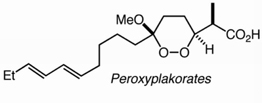
European J Org Chem. 2013 June 1; 2013(16): 3263–3270.
Abstract
A new class of twin-chain hydroxyalkylthiols (mercaptoalkanols) featuring a nearly constant cross-section and the potential for modification of one or both termini are available with complete regioselectivity through Pd-mediated couplings of benzene diiododitriflate, including an example of a previously unreported coupling to generate an ortho-substituted arene bis acetic acid. Selfassembled monolayers (SAMs) prepared from the new amphiphiles demonstrate improved stability in an electrochemical sensor system compared with monolayers prepared from analogous single chain thiols.


Comments
Copyright 2013 Fisher et al.