Chemistry, Department of: Faculty Series
Patrick Dussault Publications
Accessibility Remediation
If you are unable to use this item in its current form due to accessibility barriers, you may request remediation through our remediation request form.
Document Type
Article
Date of this Version
2012
Citation
Org Lett. 2012 May 4; 14(9): 2242–2245.
Abstract
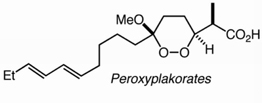
Whereas the cleavage of alkenes by ozone typically generates peroxide intermediates that must be decomposed in an accompanying step, ozonolysis in the presence of pyridine directly generates ketones or aldehydes through a process that neither consumes pyridine nor generates any detectable peroxides. The reaction is hypothesized to involve nucleophile-promoted fragmentation of carbonyl oxides via formation of zwitterionic peroxyacetals.


Comments
Copyright © 2012 American Chemical Society