Chemistry, Department of: Faculty Series
Andrzej Rajca Publications
Accessibility Remediation
If you are unable to use this item in its current form due to accessibility barriers, you may request remediation through our remediation request form.
Document Type
Article
Date of this Version
April 2001
Abstract
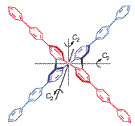
The asymmetric synthesis of a chiral, nonracemic π-conjugated system with D2 point group of symmetry, dodecaphenylene 4 is described. In the key step, (−)-sparteine- and Cu(II)-mediated oxidation of 2,2′-dilithio-1,1′-biaryls in ether gives the corresponding dimers, tetra-o-phenylenes, in 80% isolated yields and 50–60% ee’s. X-Ray crystallography confirms the structure of rac-4 and its molecular shape of a Greek cross. The torsional angles between the benzene rings in the tetra-o-phenylene core of rac-4 are in the 56.5–71.0° range. However, CD and UV spectra of 4 in CH2Cl2 are consistent with significant conjugation between the four terphenyl moieties.


Comments
Published in Tetrahedron 57:17 (April 23, 2001), pp. 3725–3735. doi:10.1016/S0040-4020(01)00241-1 http://www.sciencedirect.com/science/journal/00404020 Copyright © 2001 Elsevier Science Ltd. Used by permission.