Chemistry, Department of: Faculty Series
James M. Takacs Publications
Accessibility Remediation
If you are unable to use this item in its current form due to accessibility barriers, you may request remediation through our remediation request form.
Document Type
Article
Date of this Version
2-12-2006
Abstract
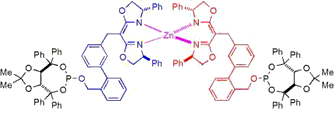
The chirality-directed self-assembly of bifunctional subunits around a structural metal-typically, zinc(II)-is used to form a heteroleptic complex in which a second set of ligating groups are suitably disposed to bind a second metal, forming a heterobimetallic catalyst system. We find that subtle changes in the structural backbone (i.e., ligand scaffold) of such chiral bidentate self-assembled ligands (SALs) can be used to manipulate the ligand topography and chiral environment around catalytic metal; thus, the scaffold can be optimized to maximize asymmetric induction. Using this combinatorial strategy for ligand synthesis, a preliminary study was carried out in which a library of 110 SALs was evaluated in the rhodium-catalyzed asymmetric hydrogenation of a simple N-acyl enamide. The level of enantioselectivity obtained varies from near racemic to greater than 80% ee as a function of the ligand scaffold, with the possibility of further improvement yet to be explored.


Comments
Published in Pure and Applied Chemistry 78:2 (February 2006), pp. 501-509. Copyright © 2006 International Union of Pure and Applied Chemistry. Used by permission.
http://www.iupac.org/publications/pac/