Chemistry, Department of: Faculty Series
James M. Takacs Publications
Accessibility Remediation
If you are unable to use this item in its current form due to accessibility barriers, you may request remediation through our remediation request form.
Document Type
Article
Date of this Version
December 2007
Abstract
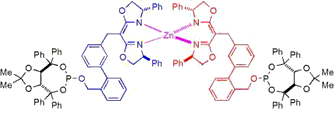
A self-assembled ligand library (SAL XY) affords a wide range of R/S ratios in Rh-catalyzed asymmetric hydroboration (nbd = 2,5norbornadiene, R* is a chiral substituent). Ligand-scaffold optimization reveals “substrate-tailored” ligands that afford high regio- and enantioselectivity for a variety of ortho-substituted styrene derivatives.
Includes article (4 pp.) and supporting information (106 pp.).


Comments
Published in Angewandte Chemie Int. Ed. 46 (2007); doi: 10.1002/anie.200703127 Online at http://www3.interscience.wiley.com/cgi-bin/abstract/117870359/ABSTRACT Copyright © 2007 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. Used by permission.